LISTEN: Quashing innovation or risking a patient’s health? Lauren Sausser told WAMU’s Health Hub on April 15 why the White House and some states are at odds over how to regulate AI in health care.
Speed, efficiency, and lower costs. Those are the traits artificial intelligence supporters celebrate. But the same qualities worry physicians who fear the technology could lead to insurance denials with humans left out of the loop.
With scant federal regulation, states are left to shape the rules on AI in health care. For residents in the Washington, D.C., metropolitan area, a divide is playing out on opposite sides of the Potomac River. Maryland and Virginia have taken very different approaches to regulating AI in health insurance.
∫⁄¡œ≥‘πœÕ¯ News correspondent Lauren Sausser joined WAMU’s Health Hub on April 15 to explain why where you live may determine how much of a role AI plays in your coverage.
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/health-industry/wamu-health-hub-ai-state-regulation-april-15-2026/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2228242&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>The federal government doesn’t track how many children have entered foster care because of immigration enforcement actions, leaving it unclear how often it happens. In Oregon, as of February two children had been placed in foster care after being separated from their parents in immigration detention cases, according to Jake Sunderland, a spokesperson for the Oregon Department of Human Services.
“Before fall 2025, this simply had never happened before,” Sunderland said.
As of mid-February, nearly by Immigration and Customs Enforcement. The record 73,000 people in detention in January represented an compared with one year before. According to , parents of 11,000 children who are U.S. citizens were detained from the beginning of Trump’s term through August.
The news outlet NOTUS that at least 32 children of detained or deported parents had been placed in foster care in seven states.
Sandy Santana, executive director of Children’s Rights, a legal advocacy organization, said he thinks the actual number is much higher.
“That, to us, seems really, really low,” he said.
Separation from a parent is deeply traumatic for children and can lead to , including post-traumatic stress disorder. Prolonged, intense stress can lead to more-frequent infections in children and developmental issues. That “toxic stress” is also associated with responsible for learning and memory, according to KFF.
, and amended existing laws during Trump’s first term to allow guardians to be granted temporary parental rights for immigration enforcement reasons. Now the enforcement surge that began after Trump returned to office last year has prompted a new wave of state responses.
In New Jersey, lawmakers are considering to amend a state law that allows parents to nominate standby, or temporary, guardians in the cases of death, incapacity, or debilitation. The bill would add separation due to federal immigration enforcement as another allowable reason.
Nevada and California passed laws last year to protect families separated by immigration enforcement actions. California’s law, called the , allows parents to nominate guardians and share custodial rights, instead of having them suspended, while they’re detained. They regain their full parental rights if they are released and are able to reunite with their children.
There are significant legal barriers to reunification once a child is placed in state custody, said Juan Guzman, director of children’s court and guardianship at the Alliance for Children’s Rights, a legal advocacy organization in Los Angeles.
If a parent’s child is placed in foster care and the parent cannot participate in required court proceedings because they are in detention or have been deported, it’s less likely they will be able to reunite with their child, Guzman said.
are U.S. citizens who live with a parent or family member who does not have legal immigration status, according to research from the Brookings Institution, a Washington, D.C.-based think tank. Within that group, 2.6 million children have two parents lacking legal status.
Santana said he expects the number of family separation cases to grow as the Trump administration continues its immigration enforcement campaign, putting more children at risk of being placed in foster care.
the agency to make efforts to facilitate detained parents’ participation in family court, child welfare, or guardianship proceedings, but Santana said it’s uncertain whether ICE is complying with those rules.
ICE officials did not respond to requests for comment for this report.
Before the change in California’s law, the only way a parent could share custodial rights with another guardian was if the parent was terminally ill, Guzman said.
If parents create a preparedness plan and identify an individual to assume guardianship of their children, the state child welfare agency can begin the process of placing the children with that individual without opening a formal foster care case, he added.
While Nevada lawmakers expanded an existing guardianship law last year to include immigration enforcement, the measure requires the parents to take the additional step of filing notarized paperwork with the secretary of state’s office, said Cristian Gonzalez-Perez, an attorney at Make the Road Nevada, a nonprofit that provides resources to immigrant communities.
Gonzalez-Perez said some immigrants are still hesitant to fill out government forms, out of fear that ICE might access their information and target them. He reassures community members that the state forms are secure and can be accessed only by hospitals and courts.
The Trump administration has taken through the Centers for Medicare & Medicaid Services, the IRS, the Supplemental Nutrition Assistance Program, the Department of Housing and Urban Development, and other entities.
Gonzalez-Perez and Guzman said that not enough immigrant parents know their rights. Nominating a temporary guardian and creating a plan for their families is one way they can prevent feelings of helplessness, Gonzalez-Perez said.
“Folks don’t want to talk about it, right?” Guzman said. “The parent having to speak to a child about the possibility of separation, it’s scary. It’s not something anybody wants to do.”
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/courts/immigrants-ice-arrests-family-separation-children-foster-care/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2178906&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>“I feel like I’m suffocating inside this shelter, trapped with no way out,” Carlos’ son said, according to one of the teens’ attorneys, when asked to describe how he felt after months at the Houston-area facility. “Every day, the same routine. Every day, feeling stuck. It makes me feel hopeless and terrified.”
During daily video calls, Carlos, who had temporary protected status, urged the siblings to be patient, to trust the process. Federal officials had vetted Carlos before he could be granted custody and told him his case was complete. He believed he would soon be back with his children, who, like him, had sought refuge from political violence in Venezuela.
An immigration officer called Carlos on a Friday and asked him to attend a meeting at an ICE office the following Monday to discuss reunification with his children. Once Carlos arrived, officers tried to force him to sign documents he said he didn’t understand. When he refused, they stripped off his clothes, seized his ID and belongings, and chained him by the neck, waist, and legs.
“They tricked me,” Carlos said in a phone call from an immigration detention center in El Paso, Texas, where he was held for several months. “They used my children to grab me,” he said.
In reporting on the family’s story, ∫⁄¡œ≥‘πœÕ¯ News reviewed court documents, spoke with the family’s immigration attorneys, interviewed Carlos, and reviewed statements from his children, translated from Spanish. Carlos is a pseudonym, being used at the request of attorneys concerned that speaking out could jeopardize Carlos’ immigration case or further delay his reunion with his family.
Using Children to Arrest Parents
Since 2003, the Department of Health and Human Services’ Office of Refugee Resettlement has cared for immigrant children under 18 who arrive in the country without their parents, often fleeing violence, abuse, or trafficking. The office, which in February had more than 2,300 children in shelters or with foster families across the country, is supposed to promptly release them to vetted caregivers, typically parents or other family members already living in the country.
Congress placed this responsibility with the health agency over 20 years ago to prioritize the well-being of unaccompanied children and separate their care from immigration enforcement priorities.
Now the second Trump administration is using migrant children held by the resettlement office to lure their parents, such as Carlos, whether or not they have a criminal record. A ∫⁄¡œ≥‘πœÕ¯ News investigation found the resettlement office, , coordinates with the Department of Homeland Security to arrest people seeking custody of migrant children.
Arrest documents show Homeland Security Investigations, the arm of the agency that normally focuses on organized criminals and traffickers, will interview parents or other caregivers then arrest them if they are in the country illegally. Before Donald Trump returned to the White House, the resettlement office prohibited data sharing and collaboration with immigration enforcement, and it did not deny caregivers custody of children solely because of their immigration status. Those last year.
It’s unclear exactly how many caregivers have been baited into arrest. LAist indicating more than 100 have been arrested while trying to get their kids out of detention, but ∫⁄¡œ≥‘πœÕ¯ News could not independently verify that number with federal agencies.
Since February, the Department of Health and Human Services, Department of Homeland Security, and Justice Department have not responded to questions about caregiver arrests. Prior to leaving DHS last month, Assistant Secretary Tricia McLaughlin said the administration protects children from being released to people who shouldn’t care for them. Andrew Nixon, an HHS spokesperson, referred questions related to immigration enforcement to DHS.
At the same time, the resettlement office has that make it harder for caregivers to gain custody of unaccompanied children. These include narrowing the range of accepted documents, requiring fingerprint-based background checks for every adult in the home and backup caregivers, and requiring in-person appointments to verify identification documents, sometimes with ICE agents present. The requirements keep “children safe from traffickers and other bad, dangerous people,” Nixon said.
As of January, the agency had detained at least 300 children already placed with vetted sponsors and asked their caregivers to reapply, according to the National Center for Youth Law and the Democracy Forward Foundation. The advocacy groups filed calling these actions “a quieter, new form of family separation.”
Reverse Separation
Dulce, a Guatemalan mother in Virginia, said her 8-year-old son was sent to a government shelter after he was detained during a traffic stop last summer while visiting family members in a different state.
At first, Dulce expected to get her son back within days — she had passed the government’s sponsorship requirements in 2024 and was reunited with him three weeks after he first crossed the border. But resettlement agency officials asked her to repeat the entire process and resubmit documents, Dulce said. It took eight months to get him back.
Dulce is a pseudonym being used at her request because she fears speaking out could get her deported.
At one point, Dulce was told to attend an interview at an ICE office to show her identification as part of the process of reuniting with her son. She refused out of fear that she too might be detained, because she doesn’t have legal status. She believes ICE agents visited her home at one point.
“I stopped going home,” Dulce said. “I lived with some of my friends for days.”
Even though she lived just 45 minutes away, Dulce was allowed to visit her son only twice a month.
Until recently, most unaccompanied children landed in government custody after being detained at the border. But border crossings started to fall in 2024, and the number of people coming to the U.S. has dropped precipitously in President Trump’s second term.
Now, hundreds of kids have been taken to government shelters after being swept up inside the country, often during immigration raids or traffic stops, according to the advocates’ lawsuit. Many were already living with relatives, including guardians already vetted by the resettlement agency.
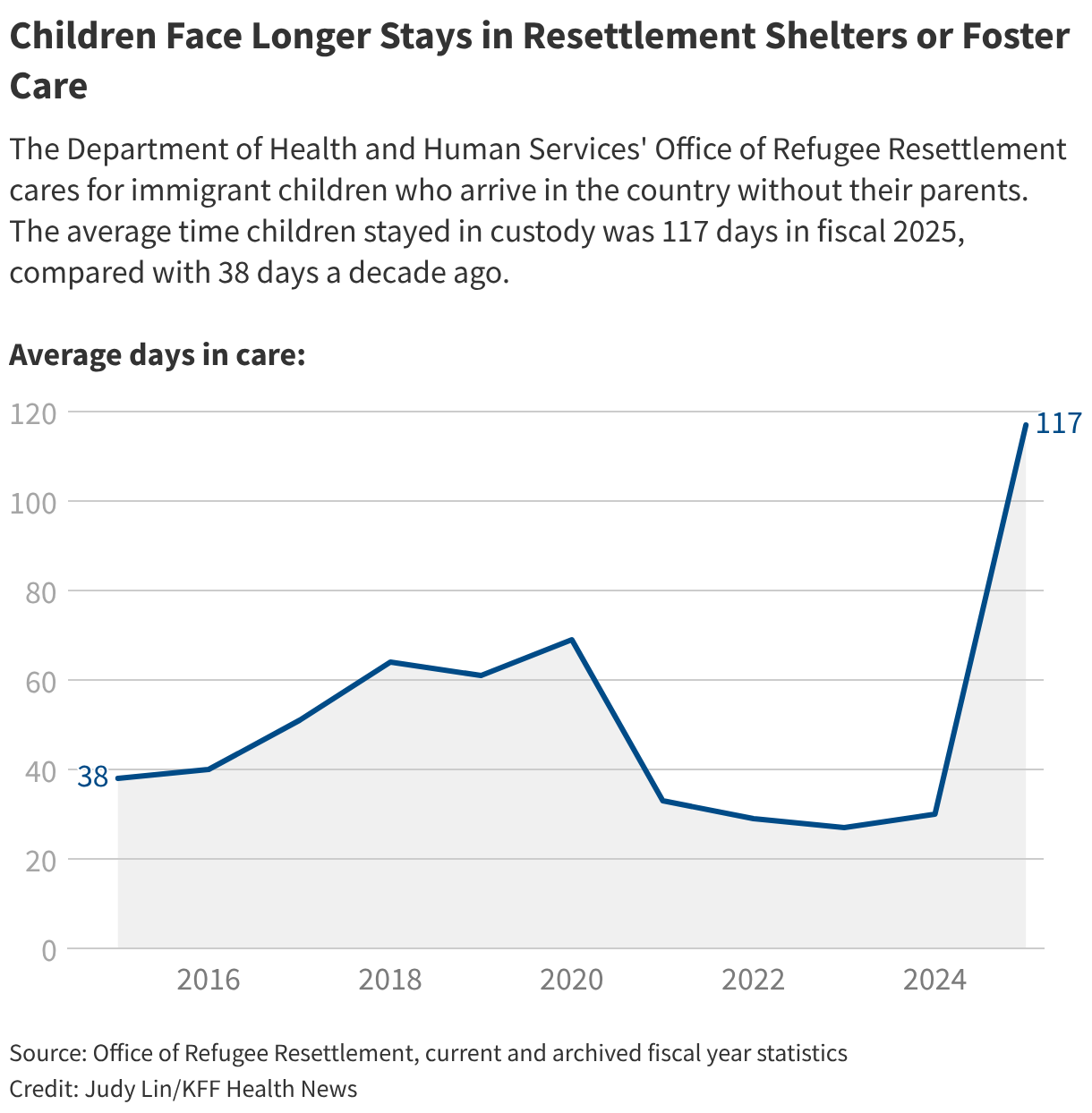
Releases have grinded nearly to a halt. According to the resettlement office, children in its custody stayed in government shelters or foster care for an average of one month in 2024. As of February, that had jumped to more than half a year.
When children do get released, it’s often only after their attorneys file a lawsuit in federal court challenging their detention as unconstitutional.
Authorities released Dulce’s son to her in February after the boy’s attorneys filed such a petition. Dulce said she’s relieved to have him back but still anxious that ICE could show up at their house.
Immigrants at Risk
During Trump’s first term, his administration was criticized for of children who had been released from custody. President Joe Biden was blamed for how his administration processed a surge of unaccompanied children that peaked in 2021 with about 22,000 in the resettlement office’s custody. Though most children were placed with legitimate sponsors, some were placed with people who hadn’t cleared , putting them at risk of .
The Trump administration says it is checking on those , and the Justice Department has prosecuted . On March 1, Homeland Security Secretary Kristi Noem, who is set to leave her role at the , touted a , including the resettlement office, that DHS said had tracked down 145,000 unaccompanied children who had been placed with caregivers during Biden’s term.
Yet internal HHS reports about that initiative obtained by ∫⁄¡œ≥‘πœÕ¯ News show that nearly 11,800 of those migrant children and nearly 500 of their caregivers were arrested as of Jan. 29. Only 125 of those migrant children and 55 of those caregivers were arrested for alleged criminal activity, suggesting the majority were for immigration violations.
HHS referred questions about the figures in the reports to DHS, which did not respond to requests for comment about the data. However, Michelle Brané, who was a DHS official in the Biden administration, said the figures show that most of the arrests were to detain and deport migrants. Previously, the administration targeted parents and caregivers who had paid for children to cross the border, trying to levy smuggling charges against them.
“They have really dropped that pretense in a lot of ways, and they are going for anyone openly,” Brané said. “These numbers clearly reflect that this is not about public safety or about safety of the children.”
Case on Hold
Carlos left Venezuela in 2022 because of death threats and, like thousands of others fleeing that country, was granted what’s called temporary protected status under the Biden administration. That protection for most Venezuelans by the Trump administration.
In January 2025, days before Trump was sworn in for his second term, Carlos’ children crossed the border from Mexico to the U.S., turned themselves over to border authorities, and were immediately placed in the resettlement agency’s custody. Carlos spent months submitting paperwork to reunite with them. He said he’s their only parent, because their mother left when they were toddlers.
Officials visited his home twice and determined he was fit to care for them, according to court documents petitioning for his release from detention. He passed DNA testing, proving he’s the biological father, one of his attorneys said. His arrest documents show he has “no criminal history.” In July, Carlos was told his reunification case was complete and being sent for approval. But then, with little explanation, the case was put on hold.
Before his arrest by ICE, Carlos said, he drove 14 hours each way from his home to visit his children. Once there, he could see them for only one hour. When he was in detention, he said, he spoke to them about every two weeks in quick, monitored phone calls.
He’s trying to stay hopeful, but it’s hard.
According to documents completed by ICE officers during his arrest and submitted in his court case, Carlos was arrested under an initiative called Operation Guardian Trace, which requires immigration officers to detain potential caregivers if they are in the country without legal authorization and recommend that they be deported.
“This operation is designed to force parents to make an impossible choice between reuniting with their children and seeking safety,” said one of Carlos’ attorneys, Chiqui Sanchez Kennedy of the Galveston-Houston Immigrant Representation Project, a nonprofit that helps low-income immigrants.
‘I’m Going to Wait’
In March, a federal judge said officials had unlawfully detained Carlos and he was released on bond.
But his children still face an uncertain future for now. Government shelters often lack sufficient resources, , and social workers say lengthy stays in these facilities can result in additional trauma.
“Not only is it bad, full stop, but the longer you’re there, the worse it gets,” said Jonathan Beier, associate director of research and evaluation for the Acacia Center for Justice’s Unaccompanied Children Program, which coordinates legal services for unaccompanied minors.
Carlos’ children could also be sent back to the country they fled. Because of his detention, Carlos will have to redo much of the process to reunite with them, according to an attorney for the children, Alexa Sendukas, also with the Galveston-Houston Immigrant Representation Project.
In statements shared through Sendukas, Carlos’ daughter said she no longer wants to be around others and spends most of the time in her room. His son, now 15, described having panic attacks and feeling that he’s missing out on life, whether it’s the opportunities he longs for — to learn English, to study science — or watching basketball with his family.

“I remember when I first arrived at this shelter, I was so hopeful and had faith that I would be reunited with my dad soon,” he said.
Carlos’ daughter spent the day crying in bed when the siblings learned their father had been detained. For days, they didn’t know where he was. Now, they fear the only way out is through adoption or foster care.
“I am afraid,” she said. “I’m going to wait for my dad forever.”
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/courts/trump-deportation-immigration-unaccompanied-children-bait-parent-arrests-hhs/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2171527&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>Raven’s case reveals a little-known gap in the U.S. donation system: There is no clear, nationally binding way to opt out ‚Äî or to ensure a later “no” overrides an earlier “yes” in a different state.
This gap, along with a range of other issues related to the organ procurement system, has become a point of bipartisan congressional concern. Late last year, the House Ways and Means subcommittee on oversight examining what members described as shortcomings, including alleged consent failures.
The panel’s scrutiny of organ procurement organizations, or OPOs, and their consent practices is a first step toward a more meaningful accountability plan that could help maintain trust across the system, according to some committee staff members.
The trust in our organ procurement and transplant system “has been eroded,” said Rep. Terri Sewell of Alabama, the panel’s senior Democrat, calling for stronger transparency and oversight to rebuild public confidence.
“Respect for autonomy ‚Äî our ability to make our own decisions (self-determination) ‚Äî allows for both ‚Äòyes’ and ‚Äòno’ decisions and for changing one’s mind,” Margaret McLean, a bioethicist at Santa Clara University, said in an email.
“Medical decision-making is not well served in a context of ambiguity,” she said.
And if a donor revokes consent, she added, “revocation by that person should carry the same ethical and procedural weight as the initial authorization, perhaps more.”
Raven Kinser Changed Her Mind
Raven was 25 when she died. Her parents, Jeff and Jaime Kinser, were at home in Michigan when they received the phone call that shattered their world. They drove through the night to the Newport News hospital, where they learned Raven’s disposition had been referred to LifeNet Health, the region’s federally designated OPO. LifeNet a failing OPO by the Centers for Medicare & Medicaid Services, meaning it doesn’t meet the government’s standards for how well it finds donors and recovers usable organs for transplant compared with other organizations.
Under federal law, hospitals are required to refer deaths and imminent deaths to OPOs, which take responsibility for donation-related decisions and discussions.
OPOs occupy a hybrid position in the health care system, as private nonprofit entities that hold exclusive, federally authorized contracts to recover organs within defined regions. They are regulated by CMS and overseen by the Health Resources and Services Administration, but that oversight occurs primarily through certification standards, performance metrics, and periodic audits rather than routine public disclosure requirements. With donor registries largely managed at the state level and no unified federal reporting requirement for removals, comprehensive national data on revocations is elusive.
OPOs are meant to separate bedside care from organ procurement decisions ‚Äî to help prevent conflicts of interest and preserve the trust that decisions about life-sustaining treatment are made solely in the dying patient’s interest. But the , leaving families unsure who is in control if and when conflicts arise.
The Kinsers, for instance, felt their daughter would not have wanted to go through the donation process, but, at the time, had no evidence. Jaime remembers telling her husband that Raven would have been mad at them for letting it happen. In an effort to stop it, Jaime inquired about whether she would be asked to sign a consent form. But a LifeNet staff member told her that wasn’t an option because donation was Raven’s “living will,” Jaime said. Meanwhile, Raven’s parents said, her personal effects, including her Virginia driver’s license, which bore no donor designation, had not yet been turned over to the family, leaving them no meaningful way to challenge LifeNet’s determination in real time.
Jaime struggled with this outcome, even mentioning in Raven’s obituary that she was an organ donor. “How would you try to make peace with something that you felt was so wrong but had no proof?” Jaime said.
Two months passed before the Kinsers gained possession of the license, which, as they had expected, showed that Raven had not opted to be an organ donor.
According to the Kinsers, LifeNet staff told them that Raven’s status as a registered donor was established by her designation on her older Michigan license.
An emailed statement attributed to Douglas Wilson, LifeNet executive vice president, said the OPO follows federal law on organ donation, the , and queries applicable state donor registries, relying on time stamps and governing law to determine the , legally valid expression of intent. Under that framework, a prior donor authorization remains enforceable unless a valid revocation is recorded in the regional OPO’s donor registry.
Because of privacy laws, Wilson said, LifeNet could not comment on the specifics of any individual case.
Raven Kinser’s choice not to be a donor when she applied for a Virginia license in July 2024 was not reflected in the registry LifeNet consulted, according to her parents, who said that is what the organization told them. According to Lara Malbon, executive director of Donate Life Virginia, which manages the state’s organ donor registry, if someone changes their donor status while completing a Virginia driver’s license or ID transaction, “that information is sent to our registry, and the registry is updated daily to reflect those changes.” Malbon also said Virginia’s registry includes only people who have “affirmatively said ‚Äòyes’ to becoming an organ, eye, and tissue donor, and it retains records solely for those who have made that decision.”
The Kinsers said they were never told why Raven’s Virginia DMV record was insufficient, or how an older yes from Michigan could outweigh a newer no in Virginia.
In December, the Kinsers filed a complaint with the Health Resources and Services Administration, urging federal regulators to investigate LifeNet’s actions and require OPOs to provide families with documented proof of the donor’s current status at the time of referral. They also called for OPOs, which operate as federally designated regional monopolies but are structured as private nonprofits, to be made subject to public records laws.
When Opting Out Doesn’t Stick
Such confusion is not unique to the Kinser family. It is a consequence of the organ donation consent process in the United States.
“I have also wondered that: why there’s not just one” registry for organ donation, Jaime said. If you go to get a firearm, you have one federal registry, she said.
Here’s how the system works: Americans typically register their organ donation intentions when they apply for driver’s licenses through state DMVs, and that decision remains governed largely by state law. That has led to 50 different sets of rules and very little federal regulation of what has become an in the U.S.
In some states, a donor checkbox is a binding legal document. In other states, the same choice may have different rules about when it takes effect, what it covers, and how it can be revoked.
Those differences can be big. State rules determine whether a person’s “gift” is limited to transplantation or also includes research and education. They determine whether the donation authorization includes tissue. And they can determine what counts as a valid revocation and when it is legally recognized.
Because of the system’s fragmentation, though, signals can cross when someone changes their mind, like Raven; it’s not always reflected from one state system to another.
Under state versions of the Uniform Anatomical Gift Act, a donor’s most recent legally valid expression of intent is meant to control.
“Personal autonomy is paramount to everything,” said Adam Schiavi, a neurointensivist who studies end-of-life decision-making. “If I say I want to be a donor, or if I say I don’t want to be a donor, that has to take precedence over everything else.”
But states differ in how revocation must be recorded and which registry is considered authoritative if someone has lived in more than one state. Those inconsistencies can create uncertainty when records conflict across jurisdictions.
“It has to be the most recent expression, not the most recent yes,” Schiavi said.
In Michigan, a change to someone’s donor status is reflected immediately in the secretary of state’s system, but only affirmative “yes” registrations appear in the registry. Removal information remains in internal motor vehicle records. In Virginia, the state registry includes only those who have affirmatively said “yes,” retaining records solely of donors, creating potential gaps if someone believes a DMV change alone is sufficient.
Elsewhere, processes and volumes differ sharply. New Mexico updates driver records in real time but does not transmit status changes to its donor registry. Instead, donor services receive restricted search access. The state logged nearly 15,000 removals in late 2021 and almost 30,000 in 2022. Florida, which maintains formal removal records through weekly DMV data files, reported 356,161 removals in 2020, more than 1.5 million in 2023, and over 1.2 million in 2025. Kentucky processed 847,371 donor registrations from 2020 to 2025, but only 16,043 icon removals, with registry withdrawal handled separately. In 2025, more than 570,000 Texans opted into the registry, while over 31,000 individuals requested removal.
According to a federal official who asked not to be identified for fear of professional repercussions, OPOs have been highly effective at lobbying states to broaden the definition of consent and authorization — shaping how those terms are applied, whether those statuses must be renewed, and how easy or difficult it is for someone to opt out.
In subsequent correspondence with federal officials, the Kinsers have urged reforms to prevent OPOs from relying on older registry entries when a more recent state DMV record exists, and they have called for criminal penalties in cases in which consent is knowingly misrepresented. Federal regulators have not indicated whether such proposals are under consideration.
Congress Takes a Closer Look
Ethicists have long cautioned that consent must be more than a checkbox and must remain grounded in respect for the donor-patient. In an October on organ transplantation, the American College of Physicians emphasized that clinicians’ primary duty is to the patient in their care, and that maintaining trust requires transparency and safeguards to prevent conflicts of interest from blurring that “bright line.”
Advocates say those steps leave unresolved the core problem raised by the Kinser family: the lack of a clear, legally binding way for people to say “no” and for that decision to follow them across state lines.
The said it “supports strengthening donor registries and enhancing registry interoperability to ensure that an individual’s documented donation decision is honored.” But OPOs have also argued that current policies protect donation as a legally enforceable gift and prevent families from overriding a loved one’s “yes” in the midst of grief. They argue that stronger, more durable consent helps reduce missed donations and saves lives.
Congress and federal regulators are considering changes to the nation’s organ donation system, including how consent is recorded and what should happen when a donor changes their mind.
Sen. Ron Wyden (D-Ore.) last year to create new federal standards for patient safety, transparency, and oversight of organ transplants, including a formal authorization for hospital or OPO staff to pause harvesting if there is any “clinical sign of life.”
HHS press secretary Emily Hilliard said the agency is “committed to holding organ procurement organizations accountable” and to “restoring integrity and transparency” to organ donation policy, calling reforms essential to informed consent and protecting donor rights. CMS issued related March 11, but it does not address the problems highlighted by the Kinsers’ case.
Critics of the organ transplant system say it is difficult for families to obtain documentation or independently verify how consent determinations were made in disputed cases.
HRSA has launched a sweeping modernization of the Organ Procurement and Transplantation Network, the national system that oversees organ allocation and transplant policy. Federal officials have described the overhaul as the most significant restructuring of the transplant system in decades, aimed at breaking up a long-standing contractor monopoly, strengthening patient safety oversight, and replacing aging technology infrastructure.
Central to that effort is modernizing the OPTN’s data systems: improving interoperability, audit trails, and transparency in how decisions are documented and reviewed. A more modern federal data architecture could make it easier to trace which registry was queried, what time stamp controlled, and how a consent determination was reached in disputed donations that span multiple states. But the modernization effort would not change the underlying state-by-state legal framework for donor authorization and what counts as a valid “no.”
Meanwhile, Donate Life America, a national nonprofit that supports state donor registries, also runs the , a central database that allows people to sign up as organ donors directly. Unlike many DMV systems, the national registry lets people log in at any time to view, update, or remove their registration and print proof of their decision. The group is also starting a project to let participating states send registrations directly into the national system, creating one place to track donor sign-ups and removals across state lines.
Each of the proposals comes with trade-offs, and both advocates and OPOs have raised concerns about how they would work in practice.
“Just doing a dump truck dump of information is not going to do much unless you really apply it through checking and auditing,” said Arthur Caplan, a professor of bioethics at New York University’s Grossman School of Medicine. “It could be like the IRS. They don’t have to audit everybody. Just do a spot audit once in a while.”
The Kinsers aren’t opposed to organ donation itself. They celebrated Raven’s donation in her obituary, and in their complaint to federal regulators, they wrote, “We are NOT anti-organ donation, and we will never take away the gift of life our oldest daughter gave to others. However, that was not LifeNet’s choice to make.”
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/health-industry/organ-donor-state-registries-consent-authorization-optn-opo-raven-kinser-virginia/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2167503&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>That’s why Ekirch, 31, of Henrico, Virginia, was stunned when she learned Anthem would finally have to pay for life-changing medical treatment.
For two years, she had battled the company to cover blood plasma infusions called intravenous immunoglobulin, or IVIG. The treatment has been shown, in some cases, to improve symptoms associated with small-fiber neuropathy, a condition that makes Ekirch’s limbs feel like they’re on fire.
But Anthem had repeatedly denied coverage for IVIG, which costs about $10,000 per infusion. Then, in February, an external review of her case conducted for the Virginia Bureau of Insurance overturned Anthem’s denial. It meant her parents would no longer need to withdraw money from her father’s retirement savings to pay out-of-pocket. Already, they’d spent about $90,000.
“My mom was sobbing. My dad was on his knees, sobbing. I don’t think I’ve ever seen him cry like that,” said Ekirch, describing her parents’ reaction to the reversal.
“I think I’m in shock from it all,” she said.
In a prepared statement, Stephanie DuBois, a spokesperson for Anthem Blue Cross and Blue Shield, said IVIG did not “align with our evidence-based standards.” But she said the company respects “the external reviewer’s decision” to overturn the denial.
Meanwhile, each year millions of patients like Ekirch continue to face denials through the prior authorization process, which requires many patients or their doctors to seek preapproval from health insurers before proceeding with medical care. And despite promises of reform from insurance companies, denials remain a frustrating hallmark of the American health care system.
Last June, Trump administration officials announced in a press conference that health insurance leaders had pledged to simplify prior authorization by taking steps such as “” subject to preapproval. The insurers also promised faster turnaround times and “clear, easy-to-understand explanations” of their decisions.
Yet in February, when ∫⁄¡œ≥‘πœÕ¯ News contacted more than a dozen major insurers that signed the pledge, half of them failed to provide specifics about health care services for which they no longer require prior authorization.
A said the industry remains committed to the effort. But physicians, consumers, and patient advocates are pessimistic about the insurers’ willingness to follow through with these voluntary changes.
“They have no desire to do what’s in the best interest of the patient if it’s going to hurt their pockets,” said Matt Toresco, CEO of Archo Advocacy, a patient advocacy and consulting company.
“In the insurance world, the fiduciary responsibility is not to the patient,” he said. “It’s to the Street,” he said, referring to Wall Street.
Meaningful Change?
The Department of Health and Human Services did not respond to questions for this article. The few updates the federal government has issued since June on prior authorization reform include a about ensuring clinicians can submit requests electronically.
AHIP, the health insurer trade group that issued the January press release, did not provide information about specific treatments, codes, medications, or procedures that its members have released from prior authorization since signing the pledge.
“We will have additional progress updates coming out later this spring,” said Kelly Parsons, a spokesperson for the Blue Cross Blue Shield Association, which represents 33 independent Blue Cross and Blue Shield companies. She also offered no specifics.
Blue Cross and Blue Shield companies that cover patients in Alabama, Arkansas, Iowa, Michigan, Pennsylvania, South Carolina, South Dakota, and Tennessee either did not respond to questions for this article or deferred to the Blue Cross Blue Shield Association.
By contrast, other insurers cited specific examples of change.
Aetna CVS Health began “bundling” prior authorizations for musculoskeletal procedures, as well as for lung, breast, and prostate cancer patients, spokesperson Phil Blando said. This practice allows providers to file one authorization request for a patient’s treatment instead of several.
And Humana removed prior authorization requirements for “diagnostic services across colonoscopies,” among other changes, spokesperson Mark Taylor said.
UnitedHealthcare, which came under intense scrutiny for its use of prior authorization following the of one of its executives in late 2024, removed prior authorization requirements on Jan. 1 for “certain nuclear imaging, obstetrical ultrasound and echocardiogram procedures,” among other changes, spokesperson Matthew Rodriguez said.
Yet some health care insiders doubt these changes will amount to much.
“Insurers have made similar promises before and failed to deliver meaningful change,” said Bobby Mukkamala, president of the American Medical Association, which represents U.S. physicians and medical students.
In 2018, , including AHIP and the Blue Cross Blue Shield Association, announced a partnership “to identify opportunities to improve the prior authorization process.” Yet, in response to the June pledge, the process remains “costly, inefficient, opaque, and too often hazardous for patients.”
“Transparency is essential so everyone can see whether real reforms are happening,” he told ∫⁄¡œ≥‘πœÕ¯ News.
Curbed Enthusiasm
Prior authorization may be getting more political attention, but data shows patients — particularly those with chronic conditions that require ongoing medical treatment — continue to face barriers to doctor-recommended care.
Among patients in that group, 39% said prior authorization is “the single biggest burden” in receiving care, according to a by KFF, a health information nonprofit that includes ∫⁄¡œ≥‘πœÕ¯ News.
I was fighting to survive, and then I was fighting to convince someone that I deserved to survive.
Anna Hocum
That’s true for Payton Herres, 25, of Dayton, Ohio, who in 2012 received a heart transplant, which requires her to take an antirejection prescription medication for the rest of her life.
But last year, she said, Anthem denied coverage for the expensive drug. She’d been taking it for more than 10 years.
“I’ve been with Anthem my entire life, and then, all of a sudden ‚Äî I don’t know what happened ‚Äî they just started denying me over and over,” she said. “I almost ran out of medication.”
DuBois, the Anthem spokesperson, confirmed the company has approved the medication. It had not taken Herres’ treatment history into account when it denied coverage for the drug, DuBois said.
But Herres said the company will require her to obtain a new authorization for the medication in September.
“Are they going to deny other things, too?” she asked. “I hope I don’t have to keep fighting like this for the rest of my life.”
Anna Hocum, 25, is preparing for a similar fight. In 2024 and 2025, her insurer repeatedly denied coverage for expensive treatment used to slow the progression of a rare genetic condition that destroys her lung function.
“I just thought I was going to die,” said Hocum, of Milwaukee. “I was fighting to survive, and then I was fighting to convince someone that I deserved to survive.”
Like with Ekirch, Hocum’s parents paid while they waited for her insurance company to overturn the initial denials. Friends and family donated more than $30,000 through a GoFundMe campaign to help defray the costs.
Then last spring, Hocum said, her insurer reversed the denial without an apparent explanation. But the approval is valid for only 12 months, so she will need another prior authorization approval this year.
“It is scary,” she said. “It’s not guaranteed that it’ll be accepted.”
They fought me tooth and nail every step of the way, to the point that they made my life a living hell.
Sheldon Ekirch
Even though it’s a “huge relief” that Anthem is now obligated to cover Ekirch’s treatment, her mother doesn’t know if or how the family will recoup the money it has already paid.
In a letter to Ekirch confirming the external reviewer’s decision, Anthem explained that the authorization would be valid for a year beginning on Sept. 25, 2025. “We are pleased we can provide a favorable response in this case,” a grievance and appeals analyst for Anthem wrote.
Ekirch said the letter highlighted the company’s hypocrisy.
“They act as though they are a benevolent organization doing me a favor.” In reality, she said, “they fought me tooth and nail every step of the way, to the point that they made my life a living hell.”
Now, Ekirch’s access to IVIG may be in jeopardy again. Her COBRA coverage through Anthem expires in late March. In April, she will need to transition to a new insurance plan ‚Äî and she’s bracing herself for another round of prior authorization.
“I just am so afraid that I don’t have the strength to go through and do what it takes,” Ekirch said, “to fight this battle again.”
Do you have an experience with prior authorization you’d like to share? to tell ∫⁄¡œ≥‘πœÕ¯ News your story.
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/health-care-costs/prior-authorization-insurer-pledge-awaiting-reforms-patients-families-bills/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2166041&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>Philip Stewart, a Rocky Mountain Laboratories researcher focused on tick-borne diseases, said he retired two years earlier than planned because of hurdles that made it too challenging to do his job well.
Alexa Romberg, an addiction prevention scientist focused on tobacco, said she “lost a great deal” of the research she oversaw when federal grants vanished.
“If one is thinking about the ‚ÄòMake America Healthy Again’ agenda and the prevention of chronic disease,” Romberg said, “tobacco use is the No. 1 contributor to early morbidity and mortality that we can prevent.”
The National Institutes of Health is the largest public funder of biomedical research in the world, with a to “enhance health, lengthen life, and reduce illness.”
Over decades, the value of the NIH may be the one thing everyone in Washington has agreed on. Lawmakers have routinely boosted its funding.
“I’m so pleased to be associated with NIH,” former Sen. Roy Blunt, a Missouri Republican and one of the NIH’s biggest champions in Congress, shortly before he retired.
But in President Donald Trump’s second term, the NIH has seen an exodus of scientists like Ernstoff, Stewart, and Romberg. Federal data shows the NIH lost about 4,400 people ‚Äî more than 20% of its workforce. Scientists say the departures harm the U.S.’ ability to respond to disease outbreaks, develop treatments for chronic illnesses, and confront the nation’s most pressing public health problems.
“People are going to get hurt,” said Sylvia Chou, a scientist who worked at the National Cancer Institute in Rockville, Maryland, for over 15 years before she left in January. “There’s going to be a lot more health challenges and even deaths, because we need science in order to help people get healthy.”
Why They’re Leaving
∫⁄¡œ≥‘πœÕ¯ News interviewed a half dozen scientists who said they quit their jobs years before they’d planned to because of the tumult of 2025.
Only a few years ago, the NIH workforce was steadily growing, from roughly 17,700 employees in fiscal year 2019 to around 21,100 in fiscal 2024, federal data shows. Under Trump, those gains have been slashed.
The Trump administration enacted a campaign to purge government workers perceived as disloyal to the president. People were fired or encouraged to leave. Officials instituted a months-long freeze on hiring.
The NIH workforce has plummeted to about 17,100 people ‚Äî its lowest level in at least two decades. Most who left weren’t fired. Roughly 4 in 5 either retired, quit, had appointments that expired, or transferred to a different job, according to federal data.

Scientists watched with dread as their colleagues were forced to terminate research funds for topics the Trump administration deemed off-limits. Across NIH labs, routine work stalled. They said they faced major delays in accessing equipment and supplies. Travel authorizations were slowed or denied.
Agency staff were instructed not to communicate with anyone outside the agency. When they could talk again, they were subject to greater constraints on what they could present to the public.
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/health-industry/nih-national-institutes-of-health-scientist-exodus-disease-treatments/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2162343&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>But when Quinn chipped a tooth and it became infected, she could not find a dentist near her home who would accept her government health coverage and was taking new patients.
She went to an emergency room, receiving painkillers and antibiotics, but she remained in agonizing pain weeks later and paid a dentist $200 to extract the tooth.
Years later, it still hurts to chew on that side, she said, but Quinn ‚Äî a 34-year-old who has four children and, with her husband, earns about $30,000 a year ‚Äî still can’t find a dentist nearby.
“You should be able to get dental care,” she said, “because at the end of the day dental care is health care.”
The federal government has long required states to offer dental coverage for children enrolled in Medicaid, the joint state-federal health program for people who are low-income or disabled. Paying for adults’ dental care, though, is optional for states.
In recent years, several states have opted to expand the coverage offered by their Medicaid programs, seeking to boost access in recognition of its importance to overall health. So far, increasing adult dental care is a work in progress: In a sampling of six of those states by ∫⁄¡œ≥‘πœÕ¯ News, fewer than 1 in 4 adults on Medicaid see a dentist at least once a year.
But under congressional Republicans’ One Big Beautiful Bill Act, which President Donald Trump signed into law last year, the federal government is expected to reduce Medicaid spending by more than $900 billion over the next decade. The range from about $184 million for Wyoming to about $150 billion for California.
State Medicaid programs typically expand or reduce benefits depending on their finances, and such massive federal cuts could force some to shrink or eliminate what they offer, including dental benefits.
“We will lose all the gains we have made,” said Shillpa Naavaal, a dental policy researcher at Virginia Commonwealth University in Richmond.
Tennessee’s Medicaid program, for instance, spent nearly $64 million on its dental coverage in 2024 and saw a 20% decrease in dental-related ER visits, said Amy Lawrence, the program’s spokesperson.
But under the new law, Tennessee is projected to lose about $7 billion in federal funding over the next decade.
As of last year, 38 states and the District of Columbia offered enhanced dental benefits for adult Medicaid beneficiaries, according to the American Dental Association. Most of the others offer limited or emergency-only care. Alabama is the only state that offers no dental coverage for adult beneficiaries.
Since 2021, 18 states have enhanced their coverage to include checkups, X-rays, fillings, crowns, and dentures, while loosening annual dollar caps for benefits.
Use of dental benefits in states with the enhanced benefits is greater than in states with only limited or emergency coverage, though still low overall, according to with the latest data as of December. No more than a third of adult Medicaid recipients saw a dentist in 2022 in any state.
To review more recent progress, ∫⁄¡œ≥‘πœÕ¯ News asked one-third of the states that have expanded their benefits in the past five years for their most recent data on the percentage of adults on Medicaid who visit a dentist at least once a year:
- Maryland — 22% (in 2024)
- Oklahoma — 16% (in 2025)
- Maine — 13% (in 2025)
- New Hampshire — 19% (in 2025)
- Tennessee — 16% (in 2024)
- Virginia — 21% (in 2025)
In comparison, about 50% to 60% of adults with private dental coverage see a dentist at least once a year, according to the ADA.
Nationwide, 41% of dentists reported participating in Medicaid in 2024, a share that has remained stable over the past decade despite the dental benefit expansions in many states, the ADA says. Many participating dentists, though, limit the number of Medicaid enrollees they treat, and some will not accept new patients on Medicaid.
Reimbursement rates have not kept up with costs, deterring dentists from accepting Medicaid, said Marko Vujicic, chief economist and vice president at the ADA Health Policy Institute.
Because of a lack of dentists who take Medicaid in southwestern Virginia, the Appalachian Highlands Community Dental Center in Abingdon sees patients who travel more than two hours for care — and must turn many away, said Elaine Smith, its executive director.
The center’s seven residents treated about 5,000 patients last year, most of them on Medicaid. About 3,000 people are on its waitlist, waiting up to a year to be seen.
“It’s sad because they have the means now to see a dentist, but they still don’t have a dental home,” Smith said.
Low-income adults face other barriers to dental care, including a lack of transportation, child care, or time off work, she said.
The inability to see a dentist has consequences broader than tooth pain. Poor dental health can contribute to a host of other significant health problems, such as heart disease . It can also make it harder to do things like apply for jobs and generally lead a healthy life.
Robin Mullins, 49, who has been off and on Medicaid since 2013, said a lack of regular dental visits contributed to her losing her bottom teeth. Unable to find a dentist near her home in rural Clintwood, Virginia, she drives almost 90 minutes to Smith’s clinic ‚Äî that is, when she can afford to get time away from driving for DoorDash or find help watching her daughter, who has special needs.
She gets by with partial dentures but misses her natural teeth, she said. “It’s absolutely horrible, as you can’t chew your food properly.”
In New Hampshire, though, the challenges have more to do with low demand than a low supply of dentists, said Tom Raffio, chief executive of Northeast Delta Dental, which manages the state’s Medicaid dental program. The company has added new dentists to its list of participating providers, along with two mobile dental units that traverse the state, he said.
Raffio said Northeast Delta Dental also has publicized the state benefits using radio advertising and social media, among other efforts.
Until 2023, New Hampshire Medicaid covered only dental emergencies.
“Culturally, it’s going to take a while,” he said, “as people just are used to not going to the dentist, or going to the ER when have dental pain.”
Brooks Woodward, dental director at Baltimore-based Chase Brexton Health Care, called Maryland’s rate of roughly 1 in 5 adults on Medicaid seeing a dentist in 2024 “pretty good” considering the benefits had been enhanced only since 2023.
Woodward said many adults on Medicaid believe that you go to a dentist only when you’re in pain. “They’ve always just not gone to the dentist, and that’s just the way they had it in their life,” he said.
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/health-care-costs/medicaid-cuts-dental-coverage-republicans-big-beautiful-bill/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2161478&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>“I was kind of a mess,” recalled Abrams, 69. Trying to cope, “I was eating myself into oblivion.” As her weight hit 270 pounds and her blood pressure, cholesterol, and blood glucose levels climbed, “I knew I was in trouble health-wise.”
What came to mind? “Oh, oh, oh, Ozempic!” ‚Äî the from television commercials that promoted the GLP-1 medication for diabetes. The ads also pointed out that patients who took it lost weight.
Abrams remembered the commercials as “joyful” and sometimes found herself humming the jingle. They depicted Ozempic-takers cooking omelets, repairing bikes, playing pickleball ‚Äî “doing everyday activities, but with verve,” she said. “These people were enjoying the hell out of life.”
So, just as such ads often urge, even though she had never been diagnosed with diabetes, she asked her doctor if Ozempic was right for her.
Small wonder Abrams recalled those ads. Novo Nordisk, which manufactures Ozempic, spent an estimated $180 million in direct-to-consumer advertising in 2022 and $189 million in 2023, according to MediaRadar, which monitors advertising.
By last year, the sum ‚Äî including radio and TV commercials, billboards, and print and digital ads ‚Äî had reached an estimated $201 million, and total spending on direct-to-consumer advertising of prescription drugs topped $9 billion, by MediaRadar’s calculations.
Novo Nordisk declined to address those numbers.
Should it be legal to market drugs directly to potential patients? This controversy, which has simmered for decades, has begun receiving renewed attention from both the Trump administration and legislators.
The question has particular relevance for older adults, who contend with more medical problems than younger people and are more apt to take prescription drugs. “Part of aging is developing health conditions and becoming a target of drug advertising,” said Steven Woloshin, who studies health communication and decision-making at the Dartmouth Institute.
The debate over direct-to-consumer ads dates to 1997, when the FDA loosened restrictions and allowed prescription drug ads on television as long as they included a rapid-fire summary of major risks and provided a source for further information.
“That really opened the door,” said Abby Alpert, a health economist at the Wharton School of the University of Pennsylvania.
The introduction of Medicare Part D, in 2006, brought “a huge expansion in prescription drug coverage and, as a result, a big increase in pharmaceutical advertising,” Alpert added. A study she co-wrote in 2023 found that pharmaceutical ads in areas with a high proportion of residents 65 and older.
and have shown that ads influence prescription rates. Patients are more apt to make appointments and request drugs, either by brand name or by category, and doctors often comply. may ensue.
But does that benefit consumers? Most developed countries take a hard pass. Only New Zealand and, despite the decadelong , the United States allow direct-to-consumer prescription drug advertising.
Public health advocates argue that such ads encourage the use and overuse of expensive new medications, even when existing, cheaper drugs work as effectively. (Drug companies don’t bother advertising once patents expire and generic drugs become available.)
In a 2023 study in JAMA Network Open, for instance, researchers analyzed the “” of the drugs most advertised on television, based on the assessments of independent European and Canadian organizations that negotiate prices for approved drugs.
Nearly three-quarters of the top-advertised medications didn’t perform markedly better than older ones, the analysis found.
“Often, really good drugs sell themselves,” said Aaron Kesselheim, senior author of the study and director of the Program on Regulation, Therapeutics, and Law at Harvard University.
“Drugs without added therapeutic value need to be pushed, and that’s what direct-to-consumer advertising does,” he said.
Opponents of a ban on such advertising say it benefits consumers. “It provides information and education to patients, makes them aware of available treatments and leads them to seek care,” Alpert said. That is “especially important for underdiagnosed conditions,” like depression.
Moreover, she wrote in a recent , direct-to-consumer ads lead to increased use not only of brand-name drugs but also of non-advertised substitutes, including generics.
The Trump administration entered this debate last September, with calling for a return to the pre-1997 policy severely restricting direct-to-consumer drug advertising.
That position has repeatedly been urged by Health and Human Services Secretary Robert F. Kennedy Jr., who has charged that “pharmaceutical ads hooked this country on prescription drugs.”
At the same time, the FDA said it was issuing about deceptive drug ads and sending “thousands” of warnings to pharmaceutical companies to remove misleading ads. Marty Makary, the FDA commissioner, in an essay in The New York Times.
“There’s a lot of chatter,” Woloshin said of those actions. “I don’t know that we’ll see anything concrete.”
This month, however, the that the agency had found its TV spot for a new oral version of Wegovy false and misleading. Novo Nordisk said in an email that it was “in the process of responding to the FDA” to address the concerns.
Meanwhile, Democratic and independent senators who rarely align with the Trump administration also have introduced legislation to ban or limit direct-to-consumer pharmaceutical ads.
Last February, independent Sen. Angus King of Maine and two other sponsors prohibiting direct-to-consumer ads for the first three years after a drug gains FDA approval.
King said in an email that the act would better inform consumers “by making sure newly approved drugs aren’t allowed to immediately flood the market with ads before we fully understand their impact on the general public.”
Then, in June, he and independent Sen. Bernie Sanders of Vermont proposed entirely. That might prove difficult, Woloshin said, given the Supreme Court’s Citizens United ruling .
Moreover, direct-to-consumer ads represent only part of the industry’s promotional efforts. Pharmaceutical firms actually spend than to consumers.
Although television still accounts for most consumer spending, because it’s expensive, Kesselheim pointed to “the mostly unregulated expansion of direct-to-consumer ads onto the web” as a particular concern. Drug sales themselves are bypassing doctors’ practices by moving online.
Woloshin said that “disease awareness campaigns” ‚Äî for everything from shingles to restless legs ‚Äî don’t mention any particular drug but are “often marketing dressed up as education.”
He advocates more effective educational campaigns, he said, “to help consumers become more savvy and skeptical and able to recognize reliable versus unreliable information.”
For example, Woloshin and Lisa Schwartz, a late colleague, designed and tested a simple “,” similar to the nutritional labeling on packaged foods, that summarizes and quantifies the benefits and harms of medications.
For now, consumers have to try to educate themselves about the drugs they see ballyhooed on TV.
Abrams read a lot about Ozempic. Her doctor agreed that trying it made sense.
Abrams was referred to an endocrinologist, who decided that her blood glucose was high enough to warrant treatment. Three years later and 90 pounds lighter, she feels able to scramble after her 2-year-old grandson, enjoys Zumba classes, and no longer needs blood pressure or cholesterol drugs.
So Abrams is unsure, she said, how to feel about a possible ban on direct-to-consumer drug ads.
“If I hadn’t asked my new doctor about it, would she have suggested Ozempic?” Abrams wondered. “Or would I still weigh 270 pounds?”
The New Old Age is produced through a partnership with .
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/aging/direct-to-consumer-advertising-big-pharma-seniors/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2157104&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>It’s all part of a drama that roiled the ACA’s 2026 open enrollment period. Congressional debate over whether to extend more generous subsidies made available under the Biden administration led to and focused public attention on rising health care costs and the affordability issue.
The enhanced subsidies, which expanded eligibility both by lowering the percentage of household income people had to pay toward their care and removing an income cap, expired at the end of last year. As a result, just about everyone buying ACA coverage saw their costs increase. For some, what they paid toward premiums doubled or more, even though less generous subsidies remain in place.
Many experts expected ACA enrollment, which hit a record 24 million in 2025, to fall this time around.
“If you raise the price of something a whole lot, economics tell us that a lot of people will buy less of it or not buy at all,” said Katherine Hempstead, a senior policy officer with the Robert Wood Johnson Foundation.
Here are things to watch now:
Initial Numbers Aren’t Final
The in December 2024 that not extending the enhanced subsidies would cause 2.2 million people to lose insurance in 2026, with further increases in following years. Analysts with the Wakely Consulting Group would opt out of insurance for this year.
Data released Jan. 28 by federal officials showed a year-over-year enrollments across the federal healthcare.gov marketplace and those run by states. Overall, there were 23 million enrollees, including 3.4 million new to ACA coverage.
At about the same time last year, there were , with 3.9 million new to the marketplaces.
But there’s more to it than those initial numbers.
For one thing, both years’ data was pegged to Jan. 15 for the federal marketplace, which closed its open enrollment period that day. But, the data for the states that run their own marketplaces included sign-ups in most cases only through Jan. 10 or 11, even though some held open enrollment until the . Thus, the numbers don’t reflect what might have happened in those last days. Was there a surge in state sign-ups? Or, conversely, did the marketplaces see more enrollees cancel their coverage?
Additionally, those initial numbers are a mix of newly minted ACA enrollees and existing customers, many of whom were auto-reenrolled for 2026 — which raises other issues.
For existing, reenrolled policyholders, the real figures won’t be known for weeks or months, when it becomes clear how many actually pay their premiums. Some consumers may not have focused on their reenrollment costs or may have hoped Congress would extend the subsidies.
That’s an important factor to keep in mind because the CBO and Wakely estimates of millions losing insurance were based on projections for full-year coverage, not initial sign-ups.
In the coming weeks, “consumers may find they really can’t afford the premiums and cancel their plans, while carriers may also cancel coverage for nonpayment,” said Pat Kelly, executive director of Your Health Idaho, a state-based ACA marketplace, during a Jan. 22 call with reporters.
Sharp Differences in State Enrollment Patterns
Changes are also afoot in the 19 other states (and the District of Columbia) that , some of which have issued more detailed data about enrollment than the federal marketplace.
Most states saw lower enrollment for 2026 than the prior year, with the biggest drop in North Carolina, where sign-ups fell by nearly 22%, federal data shows.
In a few states — including New Mexico, Texas, California, and Maryland, as well as the District of Columbia — the number of people selecting ACA plans increased.
The jump was largest in New Mexico, with its initial number of people selecting plans up by nearly 14%. Increases were in the single digits in the other states and Washington, D.C.
New Mexico — uniquely — used its own tax dollars to fully offset the loss of the more generous federal tax subsidies for all consumers. , including California, Colorado, Maryland, and Washington, used state money to help some enrollees.
The , a collective of 22 state marketplaces supported by the National Academy for State Health Policy, said initial enrollment figures . Compared with the same time last year, outright plan cancellations are up 83% in Colorado, disenrollments are four times what they were in Idaho, and Virginia has seen cancellations double.
New enrollments are from the same period last year, according to data from the state. In Pennsylvania, people ages 55 to 64, the group with the highest premiums, and young people 26 to 34 in higher numbers than other age groups, state data shows.
“We have drastically higher rates of people dropping their coverage,” said Devon Trolley, executive director of the Pennsylvania Health Insurance Exchange Authority. “We had 70,000 drop in the last two months, from early retirees to small-business owners to farmers not knowing how to make ends meet.”
On Feb. 9, Pennsylvania released , showing enrollment dropped by about 2% from last year, although that figure masks some of the effects. The state says nearly 18% of enrollees dropped coverage altogether, with older and rural residents being the most likely to fall out.
Some Republicans credited Trump-administration-backed anti-fraud measures, which included a range of , for tightening the system. Although some of those actions were paused by a federal court and have not taken effect, those ACA critics, some of whom have produced that millions may have been improperly enrolled, say that’s behind the decline. They have previously for unauthorized enrollments or ACA plan-switching by commission-seeking brokers.
States that run their own ACA marketplaces, however, reported little or no such unauthorized switching. Relative to the federal marketplace, the state-based ACA platforms employ additional safeguards to prevent brokers from accessing consumers’ coverage without authorization.
Among consumers not returning to the marketplace, the main reason is cost, said Mila Kofman, executive director of the DC Health Benefit Exchange Authority, which runs the district’s ACA marketplace.
“When we looked at who these folks are, half are small-business owners,” Kofman said. “They are not folks committing fraud.”
Lower Premiums, Higher Deductibles
Rather than sticking with automatic reenrollment, existing customers in many states shifted sharply into lower-priced “bronze” plans that come with higher deductibles than silver, gold, and platinum plans.
California saw 73% of renewing members who switched plans move to a bronze plan, up from 27% at the same time last year, the State Marketplace Network reported. In Maine, bronze enrollment now represents almost 60% of all plans purchased.
People are “looking at what works in their monthly budget, looking for that lower premium,” said Stacey Pogue, a senior research fellow at the Center on Health Insurance Reforms at Georgetown University. “Some might be crossing their fingers that they won’t need to meet their deductible.”
On average, bronze plans have an . All ACA plans are required to cover certain preventive services — such as some vaccinations, cancer screenings, and other tests — without a copayment or deductible, but most everything else is covered only after an annual deductible is met.
High deductibles can lead some patients to avoid seeking medical care, Hempstead said.
“People are terrified to use their care,” she said. “They may delay something until it’s more serious.”
She added that medical providers, including hospitals and doctors, are bracing for an increase in the number of insured patients who can’t afford to pay their deductibles.
“Everyone is anticipating that hospitals will have to give out more charity care, which will hurt their bottom lines and might lead them to have to lay off people or close or reduce services,” she said.
Are you struggling to afford your health insurance? Have you decided to forgo coverage? Click here‚ÄØto contact ∫⁄¡œ≥‘πœÕ¯ News and share your story.
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/health-care-costs/affordable-care-act-aca-obamacare-sign-ups-subsidies-higher-premiums/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2150584&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>Ballad Health announced in January that it would spend about $44 million to rebuild the 10-bed Unicoi County Hospital in a field behind a Walmart in Unicoi, Tennessee, about 7 miles from the shuttered hospital that was the site of catastrophic flooding and a daring helicopter rescue on Sept. 27, 2024.
But the new location also faces significant flood risk, according to a ∫⁄¡œ≥‘πœÕ¯ News review of information from and , two climate data companies whose flood modeling is considered more sophisticated than outdated flood maps published by the Federal Emergency Management Agency. Both Fathom and First Street estimate that a 100-year flood ‚Äî a weather event more common and less intense than Helene ‚Äî could cover much of the hospital site with more than 2 feet of water.
“The proposed site is so obviously a flood plain geomorphologically,” said Oliver Wing, chief scientific officer at Fathom. “You don’t need a model to see that.”
∫⁄¡œ≥‘πœÕ¯ News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF‚Äîan independent source of health policy research, polling, and journalism. Learn more about .This <a target="_blank" href="/health-industry/unicoi-county-hospital-tennessee-rebuild-flood-plain-risk-fema-ballad-health/">article</a> first appeared on <a target="_blank" href="">KFF Health News</a> and is republished here under a <a target="_blank" href=" Commons Attribution-NonCommercial-NoDerivatives 4.0 International License</a>.<img src="/wp-content/uploads/sites/8/2023/04/kffhealthnews-icon.png?w=150" style="width:1em;height:1em;margin-left:10px;">
<img id="republication-tracker-tool-source" src="/?republication-pixel=true&post=2152309&ga4=G-J74WWTKFM0" style="width:1px;height:1px;">]]>


